CD99, a cell surface molecule, has emerged as an essential marker for Ewing Sarcoma (EWS). [1] The disease, which expresses high levels of CD99, is the second most common bone cancer in children and young adolescent, often first appearing during the teenage years.
While Ewing sarcoma can develop in any bone, it usually affects the long bones (femur, tibia, and humerus), and in the pelvis. Less often, the tumor begins in the muscles and soft tissues of the chest, abdomen, arms or other areas, known as extraosseous (extraskeletal) Ewing’s sarcoma. Other forms of Ewing Sarcoma include peripheral primitive neuroectodermal tumor (pPNET), which occur in nerve tissue and can be found in many parts of the body, and Askin tumor, a rare type pPNET found in the chest. Ewing sarcoma can metastasize to other parts of the body, including the lungs, bone marrow, and other soft tissues. [2]
A genetic mutation
There is no known cause of Ewing sarcoma. However, a common genetic abnormality in Ewing Sarcoma involves a translocation between chromosomes 11 and 22, specifically t(11;22)(q24;q12). This translocation results in the fusion of the EWS1 gene on chromosome 22 with the FLI1 gene on chromosome 11, creating the EWS/FLI1 fusion gene. Ewing Sarcoma is not inherited but acquired during the patient’s lifetime and is only present in tumor cells. This type of mutation is known as a somatic mutation.[2]
Treatment
While aggressive multimodality therapy, which generally includes local surgery, radiotherapy and polychemotherapy, have led to improved outcomes in patients diagnosed with localized disease, therapy-related toxicity is associated with acute and chronic adverse effects that may impact the patient’s heath related Quality of Life (hrQoL). The prognosis for patients with relapsed and/or metastatic disease continues to be poor.[3] As a results there remains a major unmet medical need for patients diagnosed with Ewing Sarcoma.
Research
CD99 (also known as MIC2), a glycosylated transmembrane protein, exhibits an oncogenic function which showed strong expression levels on various cancer cell types. Over-expressed in Ewing sarcoma, CD99 contributes to the malignant behavior of cells, including their ability to proliferate, evade apoptosis, and metastasize to distant organs. Inhibiting CD99 disrupt the interaction between CD99 and its binding partners, which can include other cell surface proteins and intracellular signaling molecules. In malignant cels, this disruption can lead to the induction of apoptosis, or programmed cell death. Furthermore, inhibiting CD99 impair the adhesion properties of cancer cells, making it more difficult for them to invade surrounding tissues and metastasize. Inhibiting CD99 also inhibits migration. [4]
At NanoValent Pharmaceuticals, we have developed an investigational novel format nano-ADCs likeTargeted NanoSpheres (nADC/TNS). Based on this targeted drug delivery platform technology, we have developed NV103, a CD99 targeting nADC/TNS with an irinotecan payload which selectively binds to Ewing Sarcoma cells while largely bypassing normal, healthy, tissue and could significantly improve treatment efficacy.[5][6][7] In preclinical animal models, NV103 prolonged survival five-fold over free irinotecan treated animals in bi-weekly single dose level studies, without any treatment-related toxicity.[5]
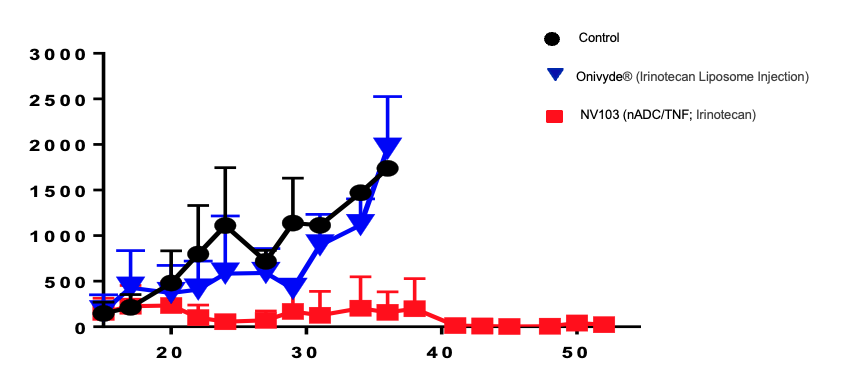
Figure 1.0. Targeted vs. Untargeted
In pre-clinical studies, NV103, our investigational novel format nano-ADCs likeTargeted NanoSpheres (nADC/TNS) targeting CD99, ablates Ewing Sarcoma. Untargeted irinotecan liposome injection (Onivyde®)* does not.
Beyond Ewing sarcoma
Our nADC/TNPs also cross the blood-brain barrier (BBB), and were used to treat orthotopic glioblastoma (GBM) PDX xenografts, prolonging animal survival 3-fold over control or free drug treated animals. Osteosarcoma, triple negative breast cancer (TNBC), glioblastoma (GBM), hepatoma, and adult Acute Lymphoblastic Leukemia (ALL) bearing animals all showed complete tumor ablation and 100% survival when treated biweekly at 10mg/kg. Pancreatic, prostate, and ovarian cancer showed 100% survival in NV103 treated mice after all control animals had died but remained tumor bearing with slow but progressive tumor growth when treated identically. Lung and colorectal cancer and melanoma bearing animals treated similarly with our nADC/TNPs showed 50-80% survival at the time of control animal death. In these preclinical studies, our team of researchers did not observe any evidence of off-target toxicity. [5]
Beyond our approach to treat Ewing Sarcoma, our unique platform technology also allowed us to develop additional investigational agents, including NV101, NV102, NV104, and NV105 to addresses the unmet medical needs of other hard to treat diseases.
Reference
[1] Manara MC, Terracciano M, Mancarella C, Sciandra M, Guerzoni C, Pasello M, Grilli A, Zini N, Picci P, Colombo MP, Morrione A, Scotlandi K. CD99 triggering induces methuosis of Ewing sarcoma cells through IGF-1R/RAS/Rac1 signaling. Oncotarget. 2016 Nov 29;7(48):79925-79942. doi: 10.18632/oncotarget.13160. PMID: 27835596; PMCID: PMC5346761.
[2] MedlinePlus [Internet]. Bethesda (MD): National Library of Medicine (US); [updated Jun 24; cited 2020 Jul 1].Genetics; Genetic Conditions, Ewing sarcoma. Online. Last accesses on April 2, 2025
[3] Grünewald TGP, Cidre-Aranaz F, Surdez D, Tomazou EM, de Álava E, Kovar H, Sorensen PH, Delattre O, Dirksen U. Ewing sarcoma. Nat Rev Dis Primers. 2018 Jul 5;4(1):5. doi: 10.1038/s41572-018-0003-x. PMID: 29977059.
[4] Scotlandi K, Baldini N, Cerisano V, Manara MC, Benini S, Serra M, Lollini PL, Nanni P, Nicoletti G, Bernard G, Bernard A, Picci P. CD99 engagement: an effective therapeutic strategy for Ewing tumors. Cancer Res. 2000 Sep 15;60(18):5134-42. PMID: 11016640.
[5] Kang H, Nagy J, Upton B, Triche T. Superior efficacy of targeted nanoparticle therapy for brain tumors [abstract]. In: Proceedings of the American Association for Cancer Research Annual Meeting 2022; 2022 Apr 8-13. Philadelphia (PA): AACR; Cancer Res 2022;82(12_Suppl): Abstract nr 295.
[6] Kang H, Upton B, Nagy J, Sarkaria JN, Triche TJ. Enhancing GBM therapy with tumor-targeted nanoparticles [abstract]. In: Proceedings of the American Association for Cancer research Annual Meeting 2025, April 25 – 30, Cgicago, Il.: AACR; 2025. Abstract nr LB183 / 1
[7] Kang H, Upton B, Nagy J, Triche T; Abstract 3206: Tumor-targeted nanoparticles for cancer therapy. Cancer Res 15 March 2024; 84 (6_Supplement): 3206.
*Onivyde® is a registered trademark of Ipsen Biopharm Ltd.

