Using our unique, highly optimized Hybrid Polymerized Liposomal Nanoparticle (HPLN) platform technology, we’re building an Industry-leading pipeline of wholly-owned drug candidates existing of our nano-ADCs likeTargeted Nanospheres (nADC/TNS).
Last updated on January 21, 2026
Our Investigational Drug Candidates
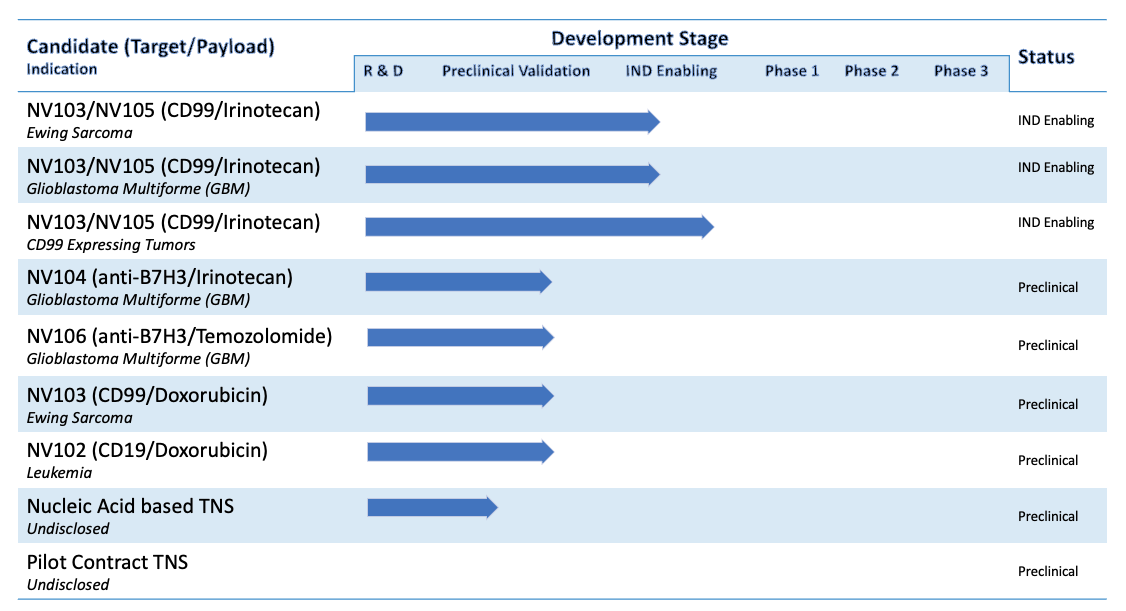
NanoValent Pharmaceuticals has developed five HPLN (Hybrid Polymerized Liposomal Nanoparticle) based nano-ADCs likeTargeted Nanospheres (nADC/TNS) candidates in oncology.
NV101 (Doxorubicin; anti-CD99)
Validating programs with NV101 in Ewing sarcoma other and CD99 expressing tumors are planned.
NV102 (Doxorubicin; anti-CD19)
Validating programs with NV102 in acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL) and other CD19 expressing tumors are planned.
NV103/NV105 (Irinotecan; anti-CD99)
NV103/NV105, our lead product, is now approaching Investigational New Drug (IND) status and is being validated in CD99-expressing tumors, including glioblastoma (GBM), ovarian cancer, pancreatic cancer, and Ewing sarcoma. (NV105; improved antibody binding).
NV104 (Irinotecan; anti-B7H3)
Validating programs with NV104 in glioblastoma (GBM) and other B7H3 (CD276) expressing tumors are planned.
NV106 (Temozolomide; anti-B7H3)
Validating programs with NV106 in glioblastoma (GBM) and and other B7H3 (CD276) expressing tumors are planned.
NV103/NV105, our investigational lead drug candidate, is now approaching U.S. Food and Drug Administration (FDA) Investigational New Drug (IND) status and is being validated in CD99-expressing tumors, including glioblastoma, pancreatic cancer, ovarian cancer, and Ewing sarcoma.
Other validating programs with NV101 in Ewing Sarcoma , NV102, in acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL), and in CD19, CD99, and CD276 (B7H3) expressing tumors are also scheduled.
- Multiple targeting antibodies: CD19, CD99 and CD276 (B7H3) antigens are preferentially express in a wide variety of tumors compared to normal, healthy, tissues
- Multiple payloads: Payloads that have successfully been delivered using our investigational novel format nano-ADCs likeTargeted NanoSpheres (nADC/TNS) include irinotecan, doxorubicin and regulatory RNAs
- Multiple cancer types: At least 75% of common cancers show dramatically improved survival in preclinical PDX mouse models when treated with NV103/NV105, our investigational lead nano-ADCs like Targeted Nanospheres (nADC/TNS)
- Combination therapy: In preclinical development, NV103/NV105 has shown to be highly effective as a single agent. Because most successful anti-cancer therapies are multi-agent based, we believe that NV103/NV105 can be successfully used as part of a combination therapy.
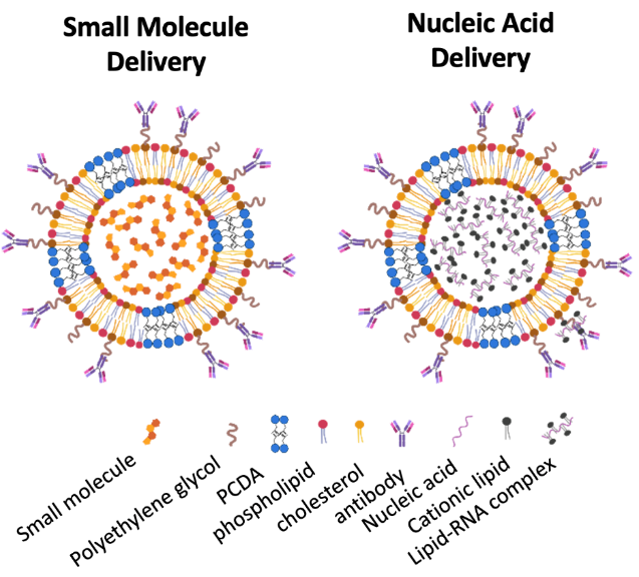
Encapsulation
Two encapsulation versions
NV103/NV105, our investigational lead drug candidate, utilizes the original aqueous interior nADC/TNS for the active-loading encapsulation of small molecules such as irinotecan.
In addition, a second-generation nADC/TNS has a solid cationic lipid interior to capture nucleic acids, with a similar shell to preserve desirable blood-brain barrier (BBB) penetration and bioavailability features of nADC/TNS.
Preclinical Development
Overall, our novel platform technology promises to significantly enhance the performance of therapeutic agents. For example, in preclinical studies, NV102 increases tumor-killing efficacy 10-12 fold in vitro compared to untargeted HPLN loaded with doxorubicin and almost 40 fold compared to conventional doxorubicin.

